Dynamic Formation of Microvillus Inclusions During Enterocyte Differentiation in Munc18-2–Deficient Intestinal Organoids
 Using intestinal organoids from Munc18-2-/- mice we show that the phenotypic manifestation of microvillus inclusion disease is critically linked to the enterocyte differentiation state. Live-cell imaging showed that microvillus inclusions gradually form either intracellularly or by plasma membrane internalization.
Using intestinal organoids from Munc18-2-/- mice we show that the phenotypic manifestation of microvillus inclusion disease is critically linked to the enterocyte differentiation state. Live-cell imaging showed that microvillus inclusions gradually form either intracellularly or by plasma membrane internalization.
Microvillus inclusion disease (MVID) is a congenital disorder that impairs nutrient absorption by the intestinal epithelial brush border. This has fatal consequences in newborn kids and there are only limited therapy options. Defective vesicular trafficking has been identified as the underlying cellular mechanism, but there is a lack of cellular models that can recapitulate this pathology. For example, intracellular microvillus inclusions (MVIs), the hallmark of this disorder, have been very rarely observed in vitro, and how these structures form remains a subject of debate.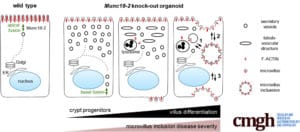
In an international collaboration Mosa and colleagues of AG Farin have now taken advantage of the 3D intestinal organoid model to study MVID. Human mutations in the MUNC18-2/STXBP2 protein cause various degrees of MVID. Using mouse mutant organoids it could now be demonstrated that the intestinal cell differentiation is a key factor for this variability. MVIs could be induced in vitro in a synchronous and highly efficiently manner, which has not been achieved in previous cellular models. By time-lapse microscopy the process of MVIs formation could be visualized for the first time. This revealed that MVIs can form in the cytoplasm or by plasma membrane invagination. The described assays and protocols could improve diagnosis of MVID using patient-derived organoids and might offer a platform to test therapeutic interventions.
Click here to directly download the paper.
Mohammed H. Mosa1,2,3, Ophélie Nicolle4,§, Sophia Maschalidi5,6,§, Fernando E. Sepulveda5,6, Aurelien Bidaud-Meynard4, Constantin Menche2, Birgitta E. Michels1,2,3,7 , Grégoire Michaux4*, Geneviève de Saint Basile5,6,8,#,* and Henner F. Farin1,2,3,#,*
1German Cancer Consortium (DKTK), Germany; 2Georg-Speyer-Haus, Institute for Tumor Biology and Experimental Therapy, Frankfurt am Main, Germany; 3German Cancer Research Center (DKFZ), Heidelberg, Germany; 4University Rennes, CNRS, IGDR UMR6290, Rennes, France; 5INSERM UMR1163, Laboratory of Normal and Pathological Homeostasis of the Immune system, Paris, France; 6Paris Descartes University-Sorbonne Paris Cité, Imagine Institute, Paris, France; 7Faculty of Biological Sciences, Goethe University Frankfurt, Germany.?8Centre d’Etudes des Déficites Immunitaires, Assistance Publique-Hôpitaux de Paris, France; § Equal contribution; #Last authors
*Corresponding Authors Grégoire Michaux: gregoire.michaux@univ-rennes1.fr | Geneviève de Saint Basile: genevieve.de-saint-basile@inserm.fr | Henner F. Farin: farin@gsh.uni-frankfurt.de
For an overview of selected publications by group leader Dr. Henner Farin, click here.
Click here for more information on Dr. Henner Farin’s research field.
Read more news about the GSH in our newsroom.



